Antibody Drug Conjugates: ‘Intelligent Drugs that Seek and Destroy Cancer’
Sep 14, 2022
What’s an ADC?
Antibody-drug conjugates, or ADCs, are highly potent biopharmaceutical drugs with unique targeting capabilities and encouraging clinical trial results. ADCs are created by linking an antibody to a biologically active drug or a cytotoxic molecule called a payload or warhead. The specificity of the antibody directs the warhead to the cancer cells. While most ADC clinical trials focus on oncology indications, ADC development is also expanding into other disease areas, such as autoimmune disorders.
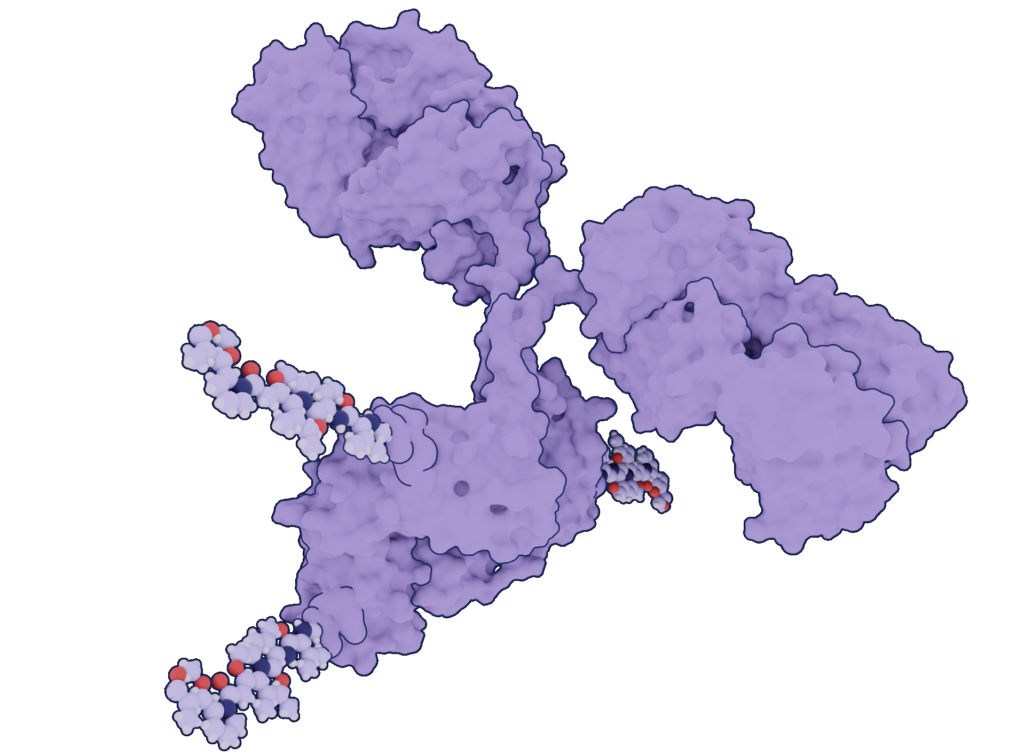
Currently, many powerful anti-cancer drugs are too toxic to be dosed on their own using a pill or injection. Because these drugs are nonspecific, they affect healthy cells along with the cancerous ones and have the potential to create severe side effects. In ADCs, the antibody provides directed delivery to cancer cells, minimizing healthy tissue’s exposure to the toxin. Once at the cancer site, the warhead is released directly into or in close proximity to the cancer cell, disrupting essential metabolic pathways required for the cancer cell to survive and essentially killing it.
The warheads are usually either small molecule drugs, radioisotopes, or peptide toxins and each antibody-drug conjugate combination has unique properties depending on the drug used and the type of antibody used as a carrier. Several types of antibodies can be used as carriers: whole antibodies, F(ab) fragments, single-chain Fv fragments, nanobodies, and more.
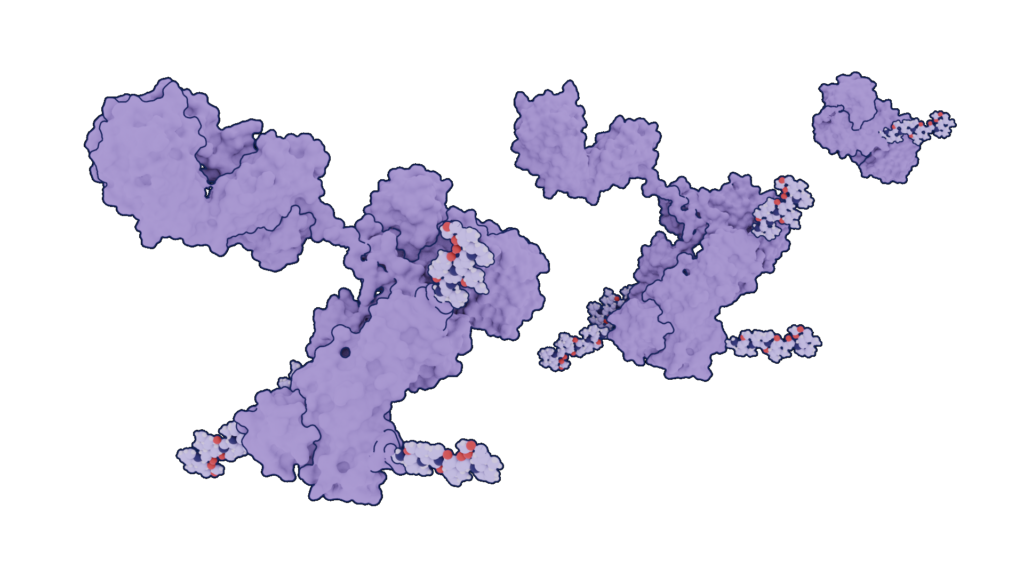
From left to right: Complete Antibody, Heavy chain fragment, and F(ab)
The cytotoxic payload/warhead can be directly coupled to the antibody or, more commonly, indirectly attached using a linker. The linkers may be non-cleavable – in such cases, the ADC must get inside cancer cells and release the toxin, acting as a Trojan horse. A linker can also be cleavable, releasing payload by enzymes or environmental differences such as pH in the tumor vicinity.
Despite the growing number of ADC approvals and applications, challenges remain in developing ADCs that demonstrate both safety and efficacy in the clinic. The research and development of ADC chemotherapy is continuing, with off-target cytotoxicity being one of the significant hurdles and concerns for clinicians.
Challenges in ADC development
Conjugation stability
One of the significant challenges of ADCs is off-target toxicity caused by the premature release of cytotoxic warheads into the bloodstream. Antibodies are giant molecules and clumsy carriers – only approximately 0.1% of ADCs reach the cancer tissue. It is then critical for the link between the antibody and the toxin to be stable to prevent damage elsewhere in the body until the drug clears out of patients’ bodies. Absci’s precise conjugation site design allows for using the most stable chemistries to secure the warhead being carried to its final destination.
Consistent Drug to Antibody (DAR) ratio
ADCs should have a consistent drug-to-antibody ratio. The most common payload to antibody stoichiometry is four drug molecules per one antibody. A constant number of linker groups improves specificity – critical characteristics of a therapeutic molecule. Minimizing conjugation reaction variability ensures the drug will meet the necessary DAR specifications, which directly impacts the potency of the ADC. Absci provides the exact number of accessible warhead conjugation points using non-standard amino acids (nsAA) incorporated into the antibody, fully controlling the DAR of each therapeutic molecule.
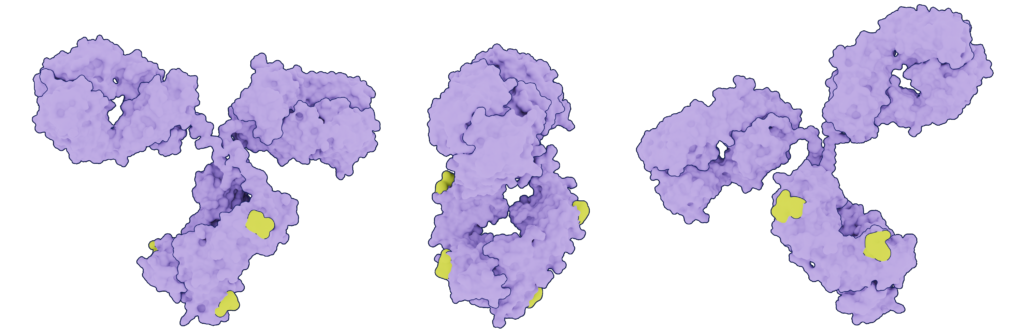
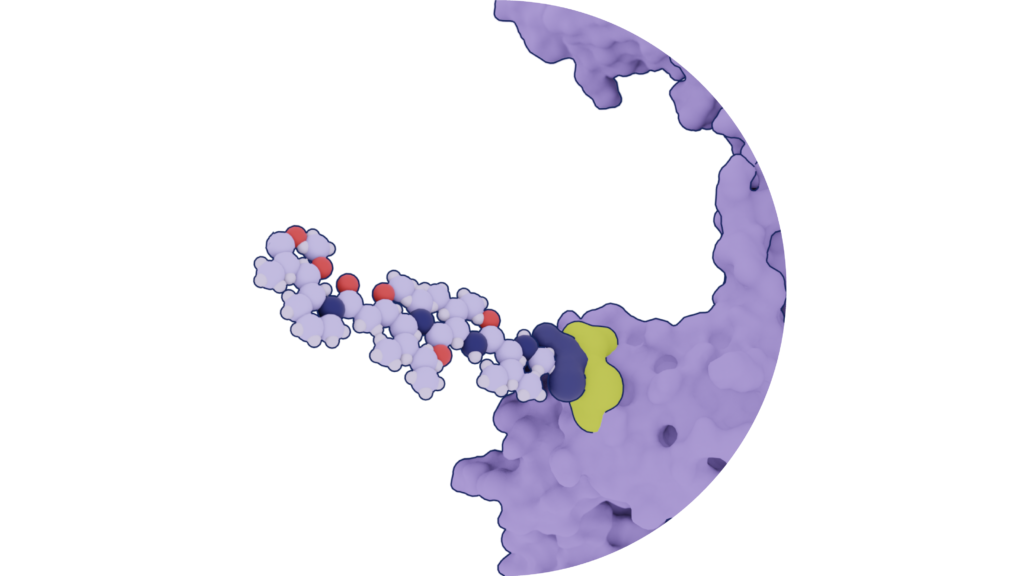
Proper positioning of the payload
In addition to achieving the optimal drug-to-antibody ratio (DAR) for each ADC, the location of the warhead on the antibody is important for therapeutic efficacy and stability. The precision of payload location can widen the therapeutic window and prevent developability troubles. To begin with, the attached warhead cannot interfere with the antibody’s binding capabilities. Also, an improper or unpredictable warhead conjugation can lead to protein aggregation. Absci can incorporate warhead conjugation points with laser precision, substituting essentially any amino acid with our nsAAs. Thus, the location of the warhead is fully determined.
Immunogenicity
Biologic drugs have an inherent risk of inducing immunogenicity, which may affect the efficacy and even be life-threatening. Antibody-drug conjugates share the same burden of immunogenicity risks as antibodies, which may jeopardize the safety and effectiveness of the drugs for patients. At Absci, we use our Naturalness score to select antibodies that are predicted to favorable characteristics, such as reduced risk off immunogenicity and developability. The score allows us to determine how close our antibodies are to the ones occurring in nature.
Let’s Work Together to Pioneer the Next Generation of ADCs
Our Bionic SoluPro™ E. coli cell line has been optimized to allow programmable site-specific incorporation of nsAAs into proteins of interest. This helps solve several key issues that plague traditional conjugation methods including high misincorporation and low homogeneity. We work with our partners on target and antibody discovery, AI-assisted antibody optimization, and site-specific payload conjugation via nsAA incorporation. Connect with us today to see how we can help you create better ADCs with precise control.